AP Biology: Elements of Life
In this AP Biology review, we will be going over learning objective 1.2.A, the elements of life.
Learning Objective
1.2.A Describe the composition of macromolecules required by living organisms.
Essential Knowledge
1.2.A.1 Atoms and molecules from the environment are necessary to build new molecules. Carbon, hydrogen, and oxygen are the most prevalent elements used to build biological molecules such as carbohydrates, proteins, lipids, and nucleic acids. Additionally:
i.Sulfur is used in the building of proteins
ii. Phosphorus is used in the building of phospholipids (a type of lipid) and nucleic
acids.
iii. Nitrogen is used in the building of nucleic acids.
Basic Chemistry
Matter is anything with mass and volume. All matter is built upon atoms, the smallest unit of matter. All atoms are built upon subatomic particles. These subatomic particles are protons, neutrons, and electrons. Here are some of the differences between the three particles:
| Particle | Mass | Charge | Location |
| Proton | 1 amu | positive | nucleus |
| Neutron | 1 amu | neutral | nucleus |
| Electron | negligible | negative | Atomic orbit |

How Elements of Life Build Macromolecules
Atoms make up elements, which are pure substances that can be broken down further. Some of the most important elements of life are carbon, hydrogen, oxygen, nitrogen, phosphorus, and sulfur. Elements are used to build macromolecules: carbohydrates, lipids, proteins, and nucleic acids.
Carbohydrates
Carbohydrates are mainly composed of carbon, hydrogen, and oxygen. Carbohydrates are composed of carbon rings and the elements always present themselves in a 1:2:1 ratio of C:H:O. For example, in glucose there are 6 carbons, 12 hydrogens, and 6 oxygens.
Lipids
Lipids are composed of carbon, hydrogen, and oxygen. Sometimes, they will contain phosphorus. For example, phospholipids contain phosphorus.
Proteins
Proteins are composed of carbon, hydrogen, oxygen, nitrogen, and sulfur. Additionally, they are composed of the following functional groups: amino group and carboxyl group. Functional groups will be discussed more in depth later.
Nucleic Acids
Nucleic Acids are made of carbon, hydrogen, oxygen, nitrogen, and phosphorus. Nucleic acids are made up of nucleotides. Nucleotides are composed of a 5-carbon sugar, a phosphate group, and a nitrogenous base.
Functional Groups
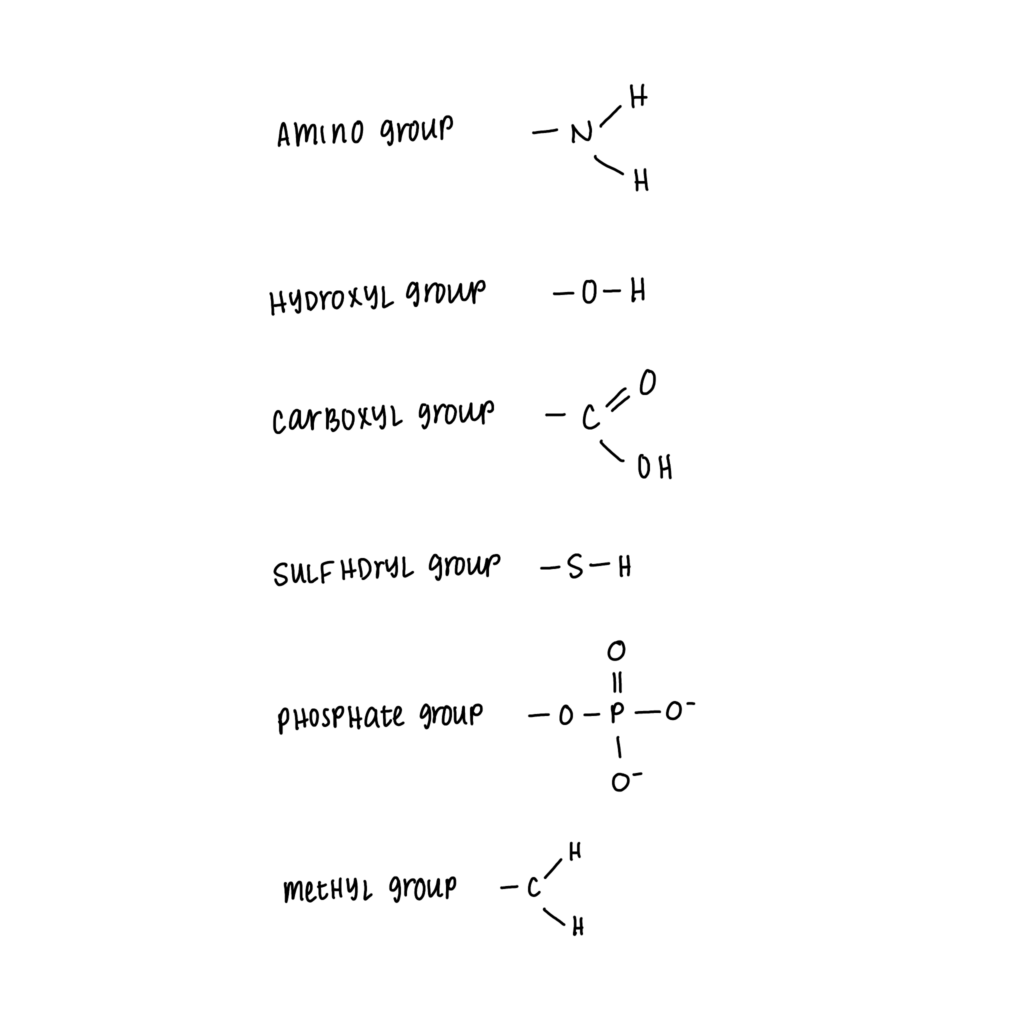
Functional groups are groups composed of specific atoms and arranged in a specific way. Functional groups can have a large influence on molecules’ properties. You do not need to know the specific properties of the functional groups, just how to identify them. There are a few you’ll need to know for the AP Biology exam.
Summary
- Atoms are made of protons, neutrons, and electrons
- Atoms –> Elements –> Macromolecules
- Macromolecules are composed of different elements:
- Carbohydrates: Carbon, hydrogen, and oxygen (CHO)
- Lipids: Carbon, hydrogen, and oxygen, sometimes phosphorous (CHOP)
- Proteins: Carbon, hydrogen, oxygen, nitrogen, and sulfur (CHOS)
- Nucleic Acids: Carbon, hydrogen, oxygen, nitrogen, and phosphorous (CHONP)
- There are 6 functional groups to know:
- Hydroxyl
- Carboxyl
- Amino
- Sulfhydryl
- Phosphate
- Methyl
If you have any questions, don’t hesitate to leave a comment! I’ll try my best to answer them! Happy studying!
-Jacqueline



